42 specific heat capacity worksheet
Specific Heat. Materials vary in their capacity to store thermal energy. For example, a material like copper will heat up much faster than water or wood. Specific Heat is a measure of the energy required to heat 1 gram of substance 1° C. Specific heat is recorded in "calories" for "mass in grams" (and "Joules for kg").
Specific heat capacity is a measurement used to quantify how good a conductor a material is. Precisely, specific heat capacity is the amount of heat required to raise one unit mass of material one degree. For example, the specific heat capacity of fresh water is 4.19 kJ/kg-K, which means that it takes 4.19 kJ of heat to raise 1 kg of water 1 K.
Copper has a specific heat capacity of 3.80 × 102 Jkg-1K-1. What temperature change would 300 g of copper experience when heated with 560 J of thermal energy? 2 ...5 pages

Specific heat capacity worksheet
A low specific heat would mean it takes very little energy to increase the temperature of the material. For example, uranium has a very low specific heat at about .116 J/(g*K). Molar Heat Capacity . Molar heat capacity, or sometimes referred to as molar specific heat, is a measure of the specific heat per unit of 1 mole of material.
Specific heat capacity worksheet with answers. A piece of copper with a mass of 218 g has heat capacity of 83 9 j c what is the specific heat capacity of copper. View answer the mass of a metal that appears to be gold is 4 30 g. Identify each variables by name the units associated with it. Identify each variables by name the units associated ...
Specific heat capacity work calculations involving specific heat specific heat calculating heat 13 0506 heat and heat calculations wkst calorimetry work w 337 name date class measuring heat transfer work answers tap 608 5 further specific and latent heat questions data specific heat capacity of.
Specific heat capacity worksheet.
A 15.75-g piece of iron absorbs 1086.75 joules of heat energy, and its temperature changes from 25°C to 175°C. Calculate the specific heat capacity of iron.
Specific heat worksheet 2 answer key 1. Specific heat is the amount of energy that it takes to raise the temperature of 1 gram of a substance by 1 degree kelvin 2. Specific heat capacity tl fi nc au296r j 7t2 tet pc6f kl ti xt how much heat is up 36 kg of hydrogen gas from 12 0 to. A 0 66 kg sample of brass at 98 6 o c is dropped into 2 33 kg ...
Specific heat may be defined as the amount of heat required to raise the temperature of one gram of a substance by 1â°c. Calculate the specific heat capacity of titanium if a 43.56 g sample absorbs 0.476 kj as its temperature changes from 20.13/c to 41.06/c. Q represents the heat added, c is the specific heat capacity of the substance you ...
Explore the meaning of calorimetry, the units of heat, specific heat capacity, and calorimetry and heat calculations. Updated: 09/07/2021 Create an account
The heat absorbed is calculated by using the specific heat of water and the equation \(\Delta H = c_p \times m \times \Delta T\). 4. Water is vaporized to steam at \(100^\text{o} \text{C}\). The heat absorbed is calculated by multiplying the moles of water by the molar heat of vaporization. 5.
Specific heat transfer image to determine the specific heat capacity of specific exam prep study sessions help, system is simply the worksheet specific heat and calorimetry problem. No isolating container is specific heat worksheet answers key for a negligible amount of reaction is a good at constant motion of solution keys.
Specific Heat Calculations Worksheet Name: Chemistry. Date: *****YOU MUST SHOW YOUR WORK TO ... Here is a chart of specific heat capacities for your use: ...
1. A 15.75-9 piece of iron absorbs 1086.75 joules of heat energy, and its temperature changes from 25°C to 175°C. Calculate the specific heat capacity of.2 pages
What is the specific heat capacity of silver metal if 55.00 g of the metal absorbs 47.3J of heat and the temperature rises 15.0°C? Page 3. 7. What mass of water ...5 pages
If the specific heat capacity of water is 4200 J/ (kg ºC), estimate the time for the temperature to rise from 10 ºC to 60 ºC. Solution: A 3 kW (3000 W) heater supplies 3000 J of heat energy per second. Let t = time taken in seconds to raise the temperature of the water by (60−10) = 50 ºC, ∴ heat supplied to water in time t seconds ...
Aug 29, 2017 - Specific Heat Capacity Worksheet - Free download as PDF File (.pdf), Text File (.txt) or read online for free. Specific Heat Capacity ...
Worksheet calculations involving specific heat. A 15 75 g piece of iron sorbs 1086 75 joules of heat energy and its temperature changes from 25 0 1750c. Heat is not the same as temperature yet they are related. Calculate the specific heat capacity of iron. Answers are provided at the end of the worksheet without units.
Specific heat chem worksheet 16 1 show all calculations for credit. Calculate the specific heat capacity of mercury. Specific heat data table 1. The mass of the water is 700 g. Engineering 032797 uploaded by. 25 0 g of mercury is heated from 25 c to 155 c and absorbs 455 joules of heat in the process. 100 0 ml of 4 0 c water is heated until its ...
Q = KA ( θ 2 - θ 1 )t/L. Where K is the coefficient of thermal conductivity of the material of the rod. K is also called thermal conductivity. Rate of flow of heat or Heat Current = H = Q/t. => H = KA ( θ 2 - θ 1 )/L. The greater the value of K for a material, the more rapidly will it conduct heat.
Specific Heat Capacity (RP) I'm a Biology teacher from the South west of England. I love Biology but I've specialised in teaching chemistry for the past 5 years. I never really got on with other resources, as I quite like to tailor everything to my own students or to my style of teaching. So I started making my own worksheets and was quite ...
Next post Specific Heat Capacity Worksheet With Answers. Related Post . Worksheet. Non Standard Measurement Worksheets Grade 1 . Adrianne Lefevre. December 8, 2021 December 8, 2021. Worksheet. Answer Key Algebra 2 Inverse Functions Worksheet ...
Waves gizmo worksheet answer key activity b. Calorie, calorimeter, joule, specific heat capacity prior knowledge questions (do these before gizmo warmup a calorimeter is an insulated container filled with a liquid, usually water. Titration gizmo answer key activity b download cult analytics. This project was created with explain everything ...
Practicing With Specific Heat Calculations Worksheet For 9th 12th Grade Lesson Planet from content.lessonplanet.com. The specific heat capacity of a substance is the amount of energy required to raise the temperature of 1 kg change of energy = m × c × change in temperature.
Specific heat capacity worksheet with answers. How many joules of heat are neeaea fo ratse the temperature of 10 0 g of. Lots of practice for using the shc equation. This covers specific heat capacity for p1 aqa. Identify each variables by name the units associated with it. View answer the mass of a metal that appears to be gold is 4 30 g.
Jan 21, 2015 — Two page worksheet using Specific Heat Capacity. Questions start easy then become gradually harder. Answers included on separate sheet. Rating: 4.7 · 66 reviews · Free · In stock
Calorie calorimeter joule specific heat capacity prior knowledge questions do these before gizmo warmup a calorimeter is. 1 calorimetry lab gizmo answer key free pdf ebook download. Will use the Calorimetry Lab Gizmo to determine the specific heat capacities. Worksheet answers student exploration calorimetry lab activity c answers ...
5) The SI unit of specific heat capacity is . a) J kg-1 K. b) J kg K. c) J kg-1 K-1. d) None. Ans: c) J kg-1 K-1 . 6) The amount of heat required to raise the temperature of 1g of water by 1°C is called as. a) 1 joule. b) 1 kJ. c) One calorie. d) None. Ans: c) one calorie . 7) The variables which describes equilibrium states of system are ...
Specific heat of water at 15oc is 1 cal/gc or 4190j/kg.k or 1 btu/lb.fo. Make sure the heat capacity t units match the thermometer t units. The common units of q are the cal (calorie) or btu (british thermal unit) 1 cal is heat required to raise the temperature of 1gm of water from 14.5 oc to 15.5oc.
In review, specific heat capacity is the amount of energy needed to change the temperature of a substance. Now, in this context, ... Quiz & Worksheet - Interspecific Competition, Competitive ...
Specific Heat and Heat of Fusion/Vaporization Notes. class 10.20-21. Click specific heat capacity problems.pptx link to view the file. ... Energy Bar Worksheet #1.. enthalpy of water, Thermodynamic properties of water: Boiling temperature (at ... 334 k J k g (the enthalpy of fusion) ⋅ 1000 J 1
Specific heat capacity worksheet key specific heat capacity. Specific heat chem worksheet 16 1 answer key as well as specific heat worksheet answers worksheet for kids maths the third section is entitled methods and treatments and it has a lot of different topics covered. How much heat energy produced this change in temperature.
Heat capacity Specific Heat Problems Worksheet. Heat Calculations Practice Flashcards Quizlet. Of heat energy and its temperature changes from 25C to 175C Calculate the gentle heat rim of iron. Calculating Specific Heat Worksheet Answers akademiexcel 092 calg C 27 Radiation convection conduction 4 Key 2020 Sample test Listening Answers We.
GCSE Physics Specific Heat Capacity Questions and Answers. Subject: Physics. Age range: 14-16. Resource type: Worksheet/Activity. (no rating) 0 reviews. Ivy2015. Last updated. 25 October 2021.
Specific heat calculations worksheet name: If the final temperature of the water was 36.4 °c, and the alloy weighed 2.71 g, what is the specific heat capacity of the alloy . 1.96 kj of heat are added to 500.
Specific heat chem worksheet 16 1 show all calculations for credit. Calculate the energy required to heat a beaker of water at 18 c to boiling. Specific Heat Chem Worksheet 16 1 Answer Key Livinghealthybulletin Best Specific Heat Che Worksheet Template Capacity Worksheets Business Plan Template Word Cal of energy is lost from a 125 […]






















![Specific Heat Capacity - Worksheet (key) [d4p7my5q264p]](https://idoc.pub/img/crop/300x300/d4p7my5q264p.jpg)

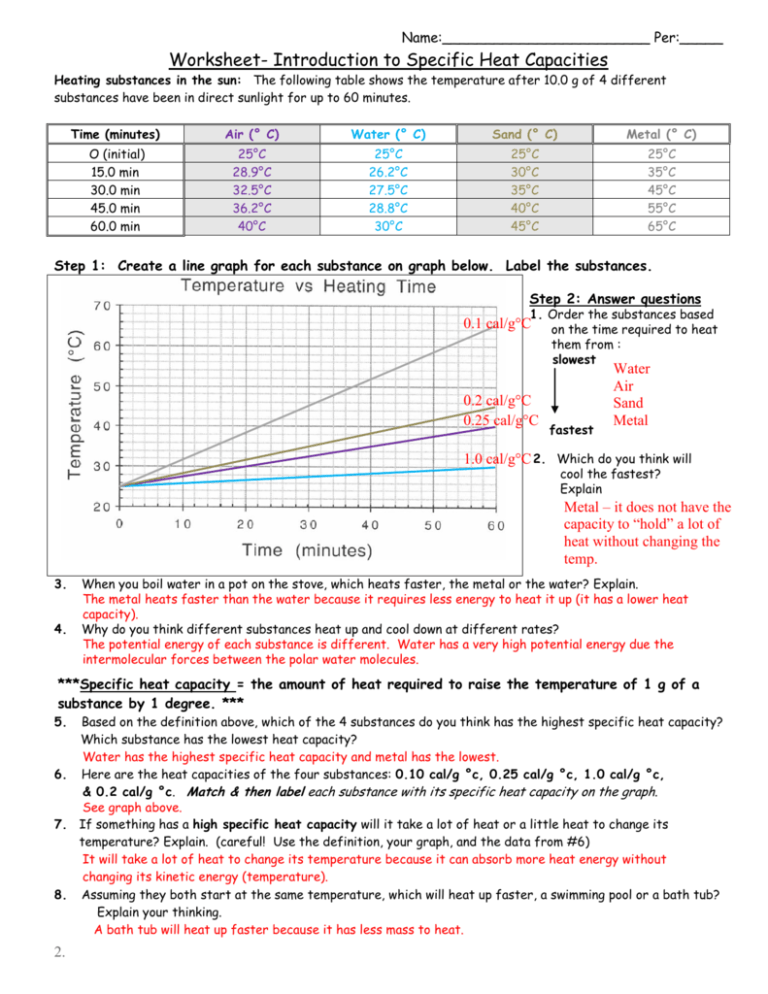














0 Response to "42 specific heat capacity worksheet"
Post a Comment