43 atomic theory review worksheet
Atom Atomic Pogil Answers Of Development The Key The Models And How have the competing explanations' experiments on atoms affected the development of the atomic model? The atomic model better represents the actual functioning of an atom first atomic theory of matter: John Dalton For example, an atom with just one proton is hydrogen, an atom with 29 protons is copper, and an atom with 94 protons is plutonium POGIL: Atomic Models and the Development of the ... DOC History of the Atom Worksheet - St. Francis Preparatory School 1. What did J.J. Thompson discover? 2. What is the charge of an electron? 3. What are cathode rays made of? 4. Why do electrons move from the negative end of the tube to the positive end? 5. What was Thompson working with when he discovered the cathode rays? Lord Ernest Rutherford (1871 - 1937):
Lesson Worksheet: Modern Atomic Theory Chemistry - Nagwa In this worksheet, we will practice describing the concepts of modern atomic theory. Q1: Who first introduced the uncertainty principle? APauli; BDe Broglie ...

Atomic theory review worksheet
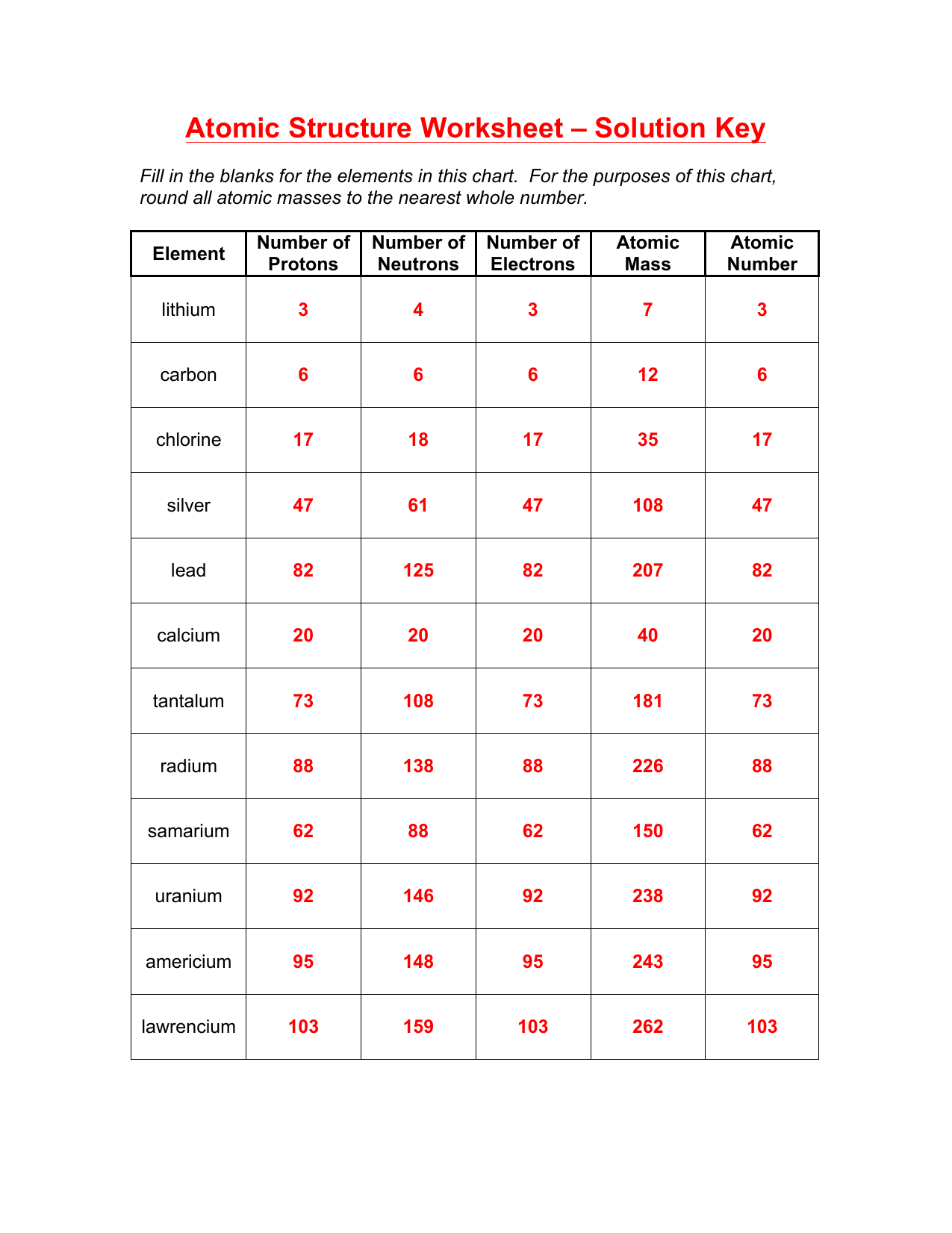
Atomic Structure Review Worksheet - Springtown ISD Atomic Structure Review Worksheet Ions 21. What is the charge of an atom with 7 protons and 9 electrons: _____ 22. What is the charge on an ion with 3 protons and 1 electron: _____ 23. What is the charge on an ion a Chlorine ion with 17 protons and 18 electrons: _____ 24. What is the charge if there are 59 protons and 62 electrons: _____ ... PDF Atomic Theory - Mr. Krohn 8th grade science - Home Development of the Atomic Theory continued What Was the First Scientific Theory of Atoms? The first scientific theory about atoms was published by John Dalton in 1803. Unlike Democritus, Dalton based his ideas on experiments. His theory helped explain observations that he and other scientists had made about elements and compounds. Atomic Theory Unit Test Review - StuDocu Atomic Theory Unit Test Review. History of Atomic Theory - Dalton: Matter's composed of small parts called atoms. - Thomson: Matter has positive and negative charged parts. - Rutherford: Positive parts (nucleus) is the center, negative parts (electrons) are in empty space, and the atom is mainly empty space. - Planck: Matter emits EM ...
Atomic theory review worksheet. Quiz & Worksheet - Atomic Theory | Study.com Worksheet. 1. The basic, modern concept of atomic theory did not develop until: The 7th century B.C. The 19th century. The 17th century. The acceptance of the Heisenberg principle. 2. Philosophers ... PDF Basic Atomic Theory, The Structure of Matter Figure 741.1.1 is a diagram of the structure of the helium atom. Notice that the nucleus is a cluster of two protons and two neutrons and that there are two electrons in an orbit, called an electron shell, around the nucleus. The atom is electrically neutral because the two positively charged protons neutralize the two negatively charged electrons. Atomic Theory - MRS. FURR'S WEBSITE Review Games AP Chemistry ATomic Theory: Unit 1. Atomic Theory Notes. Atomic Theory Worksheets. Atomic Theory Study Guide ... Atomic Theory Worksheets. Atomic Theory Study Guide. Pompom Isotope Activity. Flame lab questions. Powered by Create your own unique website with customizable templates. PDF Unit 2 Atomic Theory and Periodicity Review - WPMU DEV His theory of atomic structure led to the "plum pudding" model of atoms Thomson Electron paths cannot be defined for certain Schroedinger and Heisenberg All substances are made of atoms Dalton Section II: Atomic Vocabulary (unscramble) 1. Weighted average of all naturally occurring isotopes of the same element. (mictoa sams) atomic mass 2.
PDF The Atom for Middle School - Miss Little's Classroom Website Electron Arrangement Worksheet and Key 18. Formula Weights What You Already Know About the About the Structure of the Atom 1. Take out a piece of lined paper. 2. Number 1 to 7. 3. Write down one fact you know about the atom. 4. Turn paper over. 5. PDF CHAPTER 4 Atoms SECTION 1 The Development of Atomic Theory 3. Compare How was Dalton's atomic theory similar to Democritus's atomic theory? 4. Compare How was Thomson's atomic theory different from Rutherford's atomic theory? 5. Describe What did Thomson conclude about the particles in cathode rays? 6. Apply Concepts Nitrogen makes up 82% of the mass of ammonia. How many PDF Skills Worksheet Concept Review - whsd.net Concept Review: Energy 1. energy 2. physical 3. chemical 4. endothermic 5. exothermic 6. kinetic 7. transferred 8. In any chemical or physical change, the total quantity of energy remains constant. Energy cannot be created or destroyed. 9. Heat is the enegy transferred between objects that are at different tempera-tures. 10. Temperature is a ... Atomic Theory and Structure Review worksheet Atomic Theory and Structure ReviewAtomic Theory and Structure. ID: 2835540. Language: English. School subject: Chemistry. Grade/level: 11. Age: 16-18. Main content: Atomic Theory and Structure. Other contents: Unit Test Review. Add to my workbooks (0)
Atomic Theory Discussion worksheet 2.02 answer key.pdf Unit 2: Atomic Theory Discussion Worksheet 2 1. B. Atoms A and B are emitting photons while atoms C and D are absorbing photons. Explanation: The electron is moving from a higher energy level to a lower energy level for atoms A and B. The electron is moving from a lower energy level to a higher energy level for atoms C and D. Emission occurs when an electron moves from a higher to a lower ... PDF Honors Chem Name: Atomic Theory Exam Review Pd Atomic Theory Exam Review Pd: 1. _____He developed the solar system of the atom, although electrons do not orbit the nucleus. 2. _____ This quantum number indicates the main energy level. 3. _____When a high speed electron comes from the nucleus, what is it called? 4. 04 Atom-Review-Worksheet - Course Hero a. atoms that have the same number of protons but different numbers of neutronsb. weighted average mass of the atoms in a naturally occurring sample of an elementc. equals the number of neutrons plus the number of protonsin an atomd. 1/12the mass of a carbon-12 atom e. the number of protons in the nucleus of an atom of anelementf. an arrangement … PDF Atomic Theory - Review Sheet - Concord Consortium Atomic Theory - Review Sheet You should understand the contribution of each scientist. I don't expect you to memorize dates, but I do want to know what each scientist contributed to the theory. Democretus - 400 B.C. - theory that matter is made of atoms Boyle 1622 -1691 - defined element Lavoisier 1743-1797
Point Grey stəywəte:n̓ Secondary School Atomic Theory Worksheet No 2 (Atomic Theory Worksheet 2 Answer Key.pdf) Atomic Theory Worksheet No 3 (Atomic Theory Worksheet No 3.pdf) ... Atomic Theory Unit Review Worksheet (U6_RevWS_Key.pdf) Periodic Table. Chapter 11 Worksheet (Chapter 11 Worksheet.pdf)
DOC Quantum Theory of the Atom Worksheet Electron Configuration and Quantum Theory Worksheet. Title: Quantum Theory of the Atom Worksheet Author: rhardi Last modified by: Rosa Created Date: 9/13/2011 5:32:00 AM Other titles:
PDF AP CHEMISTRY REVIEW WORKSHEET (Unit 13 Atomic Structure and Periodicity) AP CHEMISTRY REVIEW WORKSHEET (Unit 13 - Atomic Structure and Periodicity) 1. A local radio station has a frequency of 98.6 megahertz (MHz). What is the wavelength of this station ... Account for each of the following observations in terms of atomic theory and/or quantum theory. a. Atomic size decreases from Na to Cl in the periodic table.
PDF Skills Worksheet Concept Review - whsd.net Concept Review: Energy 1. energy 2. physical 3. chemical 4. endothermic 5. exothermic 6. kinetic 7. transferred 8. In any chemical or physical change, the total quantity of energy remains constant. Energy cannot be created or destroyed. 9. Heat is the enegy transferred between objects that are at different tempera- tures. 10.
DOC Chapter 4 Review Worksheet - Currituck County Schools Greek philosopher who was among the first to suggest the existence of atoms the smallest particle of an element that retains its identity in a chemical reaction English chemist and schoolteacher who formulated a theory to describe the structure and chemical reactivity of matter in terms of atoms 1. Who did this experiment? 2. Draw in what happened?
PDF Atomic Structure Review Worksheet Key - Springtown ISD Atomic Structure Review Worksheet Key 1. 12 found below symbol on periodic table 2. Na atomic number 11 is Sodium 3. 4 mass - protons 7-3 = 4 4. 10 protons = electrons 5. 5 found by looking for the atomic number ... True 0 atomic mass units for an electron 1 atomic mass unit for protons and neutrons 15. True The electron cloud is mainly empty ...
Mr. Christopherson / Atomic Structure Worksheets *Vocabulary: Atomic Structure pdf *Atomic Number and Mass Number pdf *Ions and Subatomic Particles pdf *Development of the Atomic Theory pdf (history of the Atom paragraph) *Light Problems pdf *Half-life of Radioactive Isotopes pdf Quantum Mechanics and Electron Configuration (paragraph) *Atom, Mass, and the Mole pdf
PDF Questions for Atomic Theory Quiz #1 - Patterson Science Dalton: his atomic theory, Law of Conservation of Mass, Law of Constant Composition Atom Read Handout: In Search of a Model for Matter Read pages 23 to 25 in your text Answer questions 1 to 5 on Handout: Practice Questions: The Development of the Modern ic Theory 2 History of Atomic Theory (cont.) of the atomic models
DOC Basic Atomic Structure Worksheet - Chandler Unified School District Honors Chemistry - Unit 10 Worksheet 3. Atomic Structure. 1. Properties of Subatomic Particles. ... The atomic number gives the "identity" of an element as well as its location on the periodic table. No two different elements will have the _____ atomic number. 3. The _____ of an element is the total number of protons and neutrons in the ...
Atomic Theory Worksheet Teaching Resources | Teachers Pay Teachers Browse atomic theory worksheet resources on Teachers Pay Teachers, a marketplace trusted by millions of teachers for original educational resources. ... This cut and paste worksheet activity is the perfect way for helping your students learn and review the History of Atomic Theory. Key concepts:AtomJohn Dalton - Billiard Ball Model J.J. Thomson ...
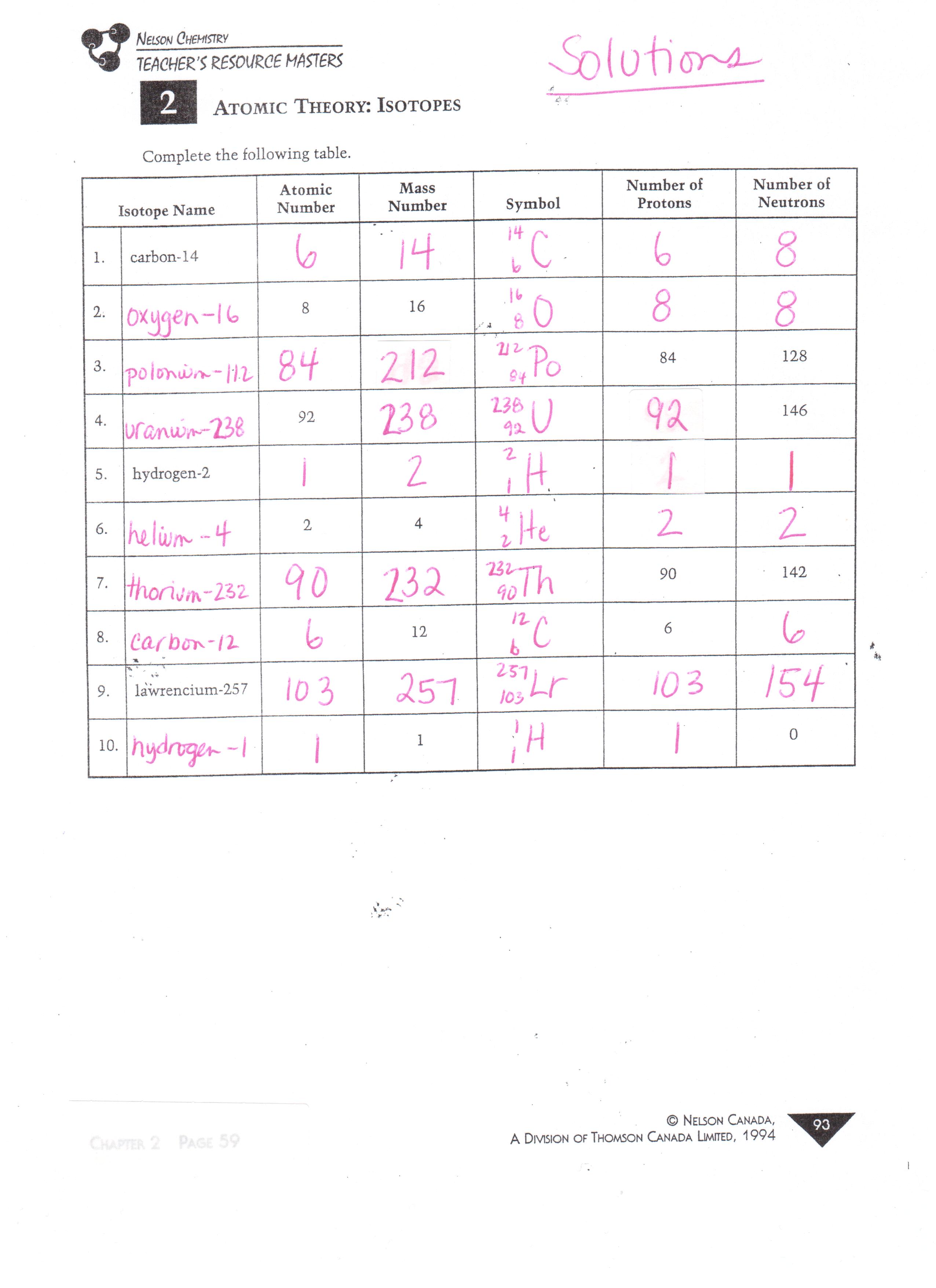
PDF Development of Atomic Theory - St. Francis Preparatory School , CHAPTER 6 REVIEW ACrlVlTY Text Reference: Section 6-90 Development of Atomic Theory Choose words from the list to fill in the blanks in the paragraphs. Word List atom atomic number Bohr Chadwick conservation of matter Dalton definite proportions electron energy level isotope Lavoisier mass number 1. multiple proportions neutron 2. nucleus
Atomic Theory Unit Test Review - StuDocu Atomic Theory Unit Test Review. History of Atomic Theory - Dalton: Matter's composed of small parts called atoms. - Thomson: Matter has positive and negative charged parts. - Rutherford: Positive parts (nucleus) is the center, negative parts (electrons) are in empty space, and the atom is mainly empty space. - Planck: Matter emits EM ...
PDF Atomic Theory - Mr. Krohn 8th grade science - Home Development of the Atomic Theory continued What Was the First Scientific Theory of Atoms? The first scientific theory about atoms was published by John Dalton in 1803. Unlike Democritus, Dalton based his ideas on experiments. His theory helped explain observations that he and other scientists had made about elements and compounds.
Atomic Structure Review Worksheet - Springtown ISD Atomic Structure Review Worksheet Ions 21. What is the charge of an atom with 7 protons and 9 electrons: _____ 22. What is the charge on an ion with 3 protons and 1 electron: _____ 23. What is the charge on an ion a Chlorine ion with 17 protons and 18 electrons: _____ 24. What is the charge if there are 59 protons and 62 electrons: _____ ...









0 Response to "43 atomic theory review worksheet"
Post a Comment