41 2.3 carbon compounds worksheet answers
2.3 Carbon Compounds | Other Quiz - Quizizz Question 3. SURVEY. 30 seconds. Q. compound made up of carbon, hydrogen, and oxygen atoms; type of nutrient that is the major source of energy for the body. answer choices. carbohydrate. lipid. protein. Biology Lesson 2.3: Carbon Compounds Guided Notes & PowerPoint Biology Lesson 2.3: Carbon Compounds Guided Notes & PowerPoint Grade Levels 8th - 12th Subjects Science, Biology, Environment Resource Type Lectures, PowerPoint Presentations, Outlines Formats Included Zip Pages 7 pages notes/20 slides $3.00 Add one to cart Buy licenses to share Add to Wish List Report this resource to TpT Room 105 Biology
PDF 2.3 Elements and Compounds - SRHS Elements and Compounds >Distinguishing Elements and Compounds Compounds can be broken down into simpler substances by chemical means, but elements cannot. 2.3 Slide 6 of 25 ... are solid carbon and water vapor. The following diagram summarizes the process. 2.3 Slide 8 of 25
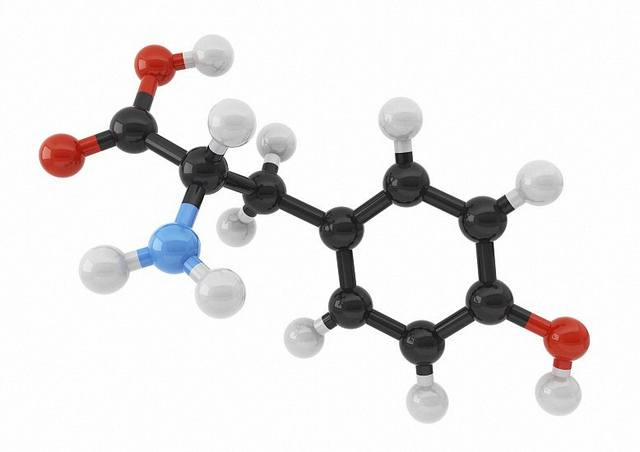
2.3 carbon compounds worksheet answers
PDF 2.3 Carbon-Based Molecules - Warren County Public Schools 2.3 Carbon-Based Molecules • Phospholipids make up all cell membranes. - Polar phosphate "head" (hydrophilic) - Nonpolar fatty acid "tails" (hydrophobic) Phospholipid 2.3 Carbon-Based Molecules 3) Proteins are polymers of amino acid monomers. - Twenty different amino acids are used to build proteins in organisms. 2.3 Carbon-Based Molecules 2.3 Carbon Compounds Flashcards | Quizlet Biology Section 2.3 Carbon Compounds. 43 terms. Makayla_Hayes51. OTHER SETS BY THIS CREATOR. 18.3 Building The Tree of Life. 12 terms. sierraboles2022. 18.2 Modern Evolutionary Classification. 22 terms. sierraboles2022. 18.1 Finding Order in Diversity. 22 terms. sierraboles2022. Classification Chapter 18 Vocabulary. 2-3 GR Worksheet Carbon Compounds.doc - Course Hero Name Class Date Section 2-3 Carbon Compounds (pages 44-48) 1.What gives carbon the ability to form chains that are almost unlimited in length? 2.Many of the molecules in living cells are so large that they are known as ______________________. 3. What is the process called by which macromolecules are formed?4.
2.3 carbon compounds worksheet answers. Chapter 2: Lesson 2.3 "Carbon Compounds" Flashcards - Quizlet Biology Section 2.3 Carbon Compounds. 43 terms. Makayla_Hayes51. Bio 2.3 Carbon Compounds. 32 terms. Brenda23Maruki. Upgrade to remove ads. Only $2.99/month. Biology - (2.3 Carbon Compounds Powerpoint and Guided Notes) The powerpoint and guided notes correspond to Section 2-3: Carbon Compounds from Miller and Levine. This powerpoint and the Guided Notes could easily be adapted to fit any biology textbook. - The powerpoint for this section is 29 slides and the guided notes are 5 pages. - Includes Daily Objectives. - Guided Notes are a Modified Cornell Note format. PDF Section 2.3 2.3 Chemical Properties - Physical Science Most candles are made from paraffin, which is a mixture of com- pounds containing carbon and hydrogen. As a candle burns, the compounds combine with oxygen in the air to form water and carbon dioxide.(Carbon dioxide is the gas that gives a carbonated beverage its fizz.) The ability to burn is a chemical property. Achemical property PDF 2.3 Carbon Compounds - isd2135.k12.mn.us Follow the directions. Then answer the questions. 1. Look at the diagram of the general structure of an amino acid. Color the amino group green. 2. Color the carboxyl group blue. 3. Color the R group red. 4. Color the same groups in the amino acids alanine and serine. 5. How many oxygen atoms are found in the carboxyl group? A. 1 C. 3 B. 2 D. 4 6.
PDF Name: Chemistry 20 Worksheets Chemistry 20 Worksheets 9 Worksheet 1.5: More difficult mole problems 1. How many atoms of copper are in 0.088 mol of copper (I) oxide? 2. How many mol of magnesium ions are in 1.00 x 1020 formula units of magnesium nitride? 3. What is the mass of 14.6 L of carbon monoxide at STP? 4. How many atoms of xenon are in 15 L at SATP? 5. PDF 2.3 - Carbon Compounds - Quia 2.3 - Carbon Compounds Regents Biology! 2.3 - Carbon Compounds Regents Biology! Carbon has 4 valence electrons which can bond covalently with other atoms can bond with other carbon atoms to form long chains can form single, double, or triple bonds… …can form a great variety of giant and complex molecules Regents Biology! PDF 2.3 Elements and Compounds - Henry County Schools ProcedureMake signs with the names of each of these items: 1 oxygen, 2 neon, 3 apple, 4 sand, 5 iron, 6 water, 7 air, 8 paint, 9 sodium chloride, 10 sucrose, 11 carbon dioxide, 12 granite, 13 laun- dry detergent, 14 citric acid, 15 cereal, 16 salad, 17 salad dressing, 18 copper, 19 ocean water, 20 gold. PDF Lesson 2.4: Physical Science Chemical Compounds - Literacy Minnesota TEACHER ANSWER KEY 1. a 2. b 3. Answers may vary, but should be similar to: They both contain the atoms of two or more elements. 4. Answers may vary, but should be similar to: A compound contains two or more elements that have combined chemically to form molecules. Mixtures contain two or more substances that have not combined chemically. 5.
PDF The Chemistry of Life - Weebly SAMPLE ANSWER: Organisms use carbon compounds to form four types of molecules: lipids, carbohydrates, nucleic acids, and proteins. SAMPLE ANSWER: A lot of what that happens in an organism is based on chemical reactions. SAMPLE ANSWER: Enzymes are proteins that speed up chemical reactions that take place in cells. PDF Chapter 2 The Chemistry of Life Worksheets Table2.1: Types of Organic Compounds TypeofCompound Examples Elements Functions Carbohydrates sugars,starches carbon,hydrogen,oxy-gen providesenergytocells, stores energy, forms bodystructures Lipids fats,oils carbon,hydrogen,oxy-gen stores energy, forms cellmembranes,carries messages Proteins enzymes,antibodies carbon,hydrogen,oxy-gen,nitrogen ... Biology 2010 Student Edition - GradeSaver Biology 2010 Student Edition answers to Chapter 2, The Chemistry of Life - 2.1 - The Nature of Matter - 2.1 Assessment - Page 38 3b including work step by step written by community members like you. ... Assessment - 2.3 Carbon Compounds - Understand Key Concepts/Think Critically; Assessment - 2.4 Chemical Reactions and Enzymes - Understand Key ... Chapter 2Properties of Matter Section 2.3 Chemical Properties - Quia 1. Is the following sentence true or false? The substances in paraffin do not change when a candle burns. 2. Circle the letters of the compounds formed when a candle burns. a.paraffin b.hydrogen c.water d.carbon 3. What is a chemical property? 4. Is the following sentence true or false? Flammability is a material's ability to burn
PDF Worksheet 2 Answers - butane.chem.uiuc.edu Worksheet 2 - Atoms, Molecules andtheir Names You must memorize the names and charges in tables 2.3-2.8. Use your Periodic Table to answer the following questions. What is the name of each of the following Groups? Group I Group Il Group h Group VIII The Group in the middle The Period 3 Halogen is The Period 2 Alkaline Earth Metal is
PDF seCTion 2.3 Carbon-Based Molecules - Weebly 2.3seCTion Carbon atoms have unique bonding properties. Most molecules that make up living things are based on carbon atoms. The structure of a carbon atom allows it to form up to four covalent bonds. It can bond to other carbons or to different atoms.
Biology 2010 Student Edition Chapter 2, The Chemistry of Life ... Biology 2010 Student Edition answers to Chapter 2, The Chemistry of Life - Assessment - 2.1 The Nature of Matter - Understand Key Concepts/Think Critically - Page 56 4 including work step by step written by community members like you. Textbook Authors: Miller, Kenneth R.; Levine, Joseph S., ISBN-10: 9780133669510, ISBN-13: 978--13366-951-0, Publisher: Prentice Hall
Worksheet_1_Bonds_Carbon_Fall20.pdf - Check your... View Worksheet_1_Bonds_Carbon_Fall20.pdf from BIO 311C at University of Texas. Check your understanding HW worksheet 1: Topics: Chemical bonds and carbon compounds Start here: Chemistry for biology
PPTX Chapter 2.3: Carbon Compounds - Fort Bend ISD Notes: Chapter 2.3Carbon Compounds Chemistry of Carbon Carbon can form four covalent bonds.(tetravalence) Carbon usually bonds with other non metals 6 protons 6 neutrons 6 electrons first shell- 2 second shell- 4 Carbon atoms have unique bonding properties. Carbon forms covalent bonds with up to four other atoms, including other carbon atoms.
PDF Worksheet 2 - University of Illinois Urbana-Champaign WORKSHEET 2 - Atoms, Molecules and Names 8. Decide if each of the following is ionic or covalent, and fill in the missing information. Name Formula ionic or covalent SF2 mercury(II) phosphate CaS diphosphorus decachloride carbon monoxide 9. Decide what is wrong with the following names, then give the correct name and formula. Name Formula ...
PPTX Biology Chapter 2- The Chemistry of Life 2.3 Carbon Compounds. What elements does carbon bond with to make up life's molecules? Carbon can bond w/ many elements, including hydrogen, oxygen, phosphorus, sulfur, carbon, and nitrogen to form molecules of life. Carbon has the ability to form millions of different large and complex structures.
PDF 2.1 The Nature of Matter - valentinebiology.weebly.com 2.3 Carbon Compounds Lesson Objectives Describe the unique qualities of carbon. Describe the structures and functions of each of the four groups of macromolecules. BUILD Vocabulary A. The chart below shows key terms from the lesson with their definitions. Complete the chart by writing a strategy to help you remember the meaning of each term.
Chapter 2.3-2.4 Test | Other Quiz - Quizizz 10 Questions Show answers Question 1 30 seconds Q. Carbon is unique due to the carbon atom's answer choices bonding properties six outer unpaired electrons ionic compounds hydrogen bonding strength Question 2 30 seconds Q. Which category of carbon-based molecules includes sugars and starches? answer choices unsaturated fatty acids phospholipids
AQA GCSE 9-1 CHEMISTRY UNIT 2.2 & 2.3 Properties ionic, covalent ... Resource type: Worksheet/Activity AQA GCSE 9-1 CHEMISTRY UNIT 2.2 & 2.3 Properties ionic, covalent, polymers, states, carbon, alloys 4.2.2 How bonding and structure are related to the properties of substances 4.2.2.1 The three states of matter 4.2.2.2 State symbols 4.2.2.3 Properties of ionic compounds 4.2.2.4 Properties of small molecules
Chapter 2 2.3 Elements and Compounds > 37 Symbols and Latin Names for Some Elements Name Symbol Latin name Sodium Na natrium Potassium K kalium Antimony Sb stibium Copper Cu cuprum Gold Au aurum Silver Ag argentum Iron Fe ferrum Lead Pb plumbum Tin Sn stannum Interpret Data
2-3 GR Worksheet Carbon Compounds.doc - Course Hero Name Class Date Section 2-3 Carbon Compounds (pages 44-48) 1.What gives carbon the ability to form chains that are almost unlimited in length? 2.Many of the molecules in living cells are so large that they are known as ______________________. 3. What is the process called by which macromolecules are formed?4.
2.3 Carbon Compounds Flashcards | Quizlet Biology Section 2.3 Carbon Compounds. 43 terms. Makayla_Hayes51. OTHER SETS BY THIS CREATOR. 18.3 Building The Tree of Life. 12 terms. sierraboles2022. 18.2 Modern Evolutionary Classification. 22 terms. sierraboles2022. 18.1 Finding Order in Diversity. 22 terms. sierraboles2022. Classification Chapter 18 Vocabulary.
PDF 2.3 Carbon-Based Molecules - Warren County Public Schools 2.3 Carbon-Based Molecules • Phospholipids make up all cell membranes. - Polar phosphate "head" (hydrophilic) - Nonpolar fatty acid "tails" (hydrophobic) Phospholipid 2.3 Carbon-Based Molecules 3) Proteins are polymers of amino acid monomers. - Twenty different amino acids are used to build proteins in organisms. 2.3 Carbon-Based Molecules





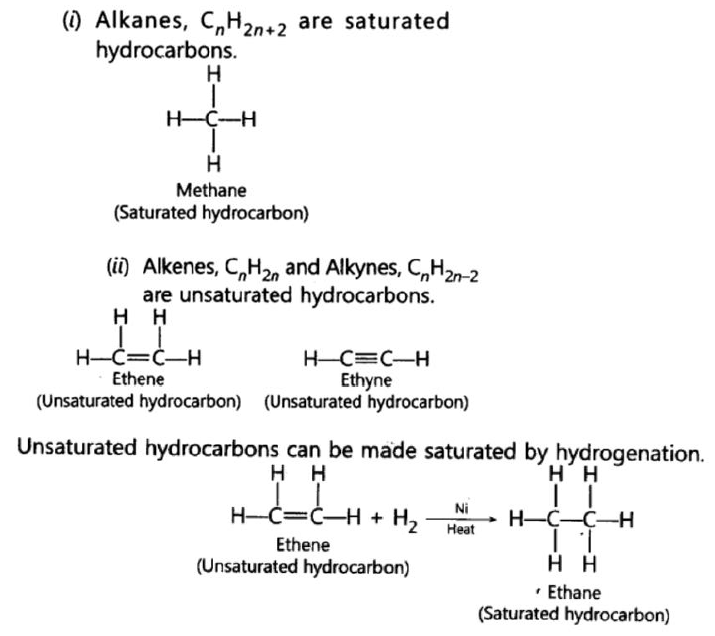







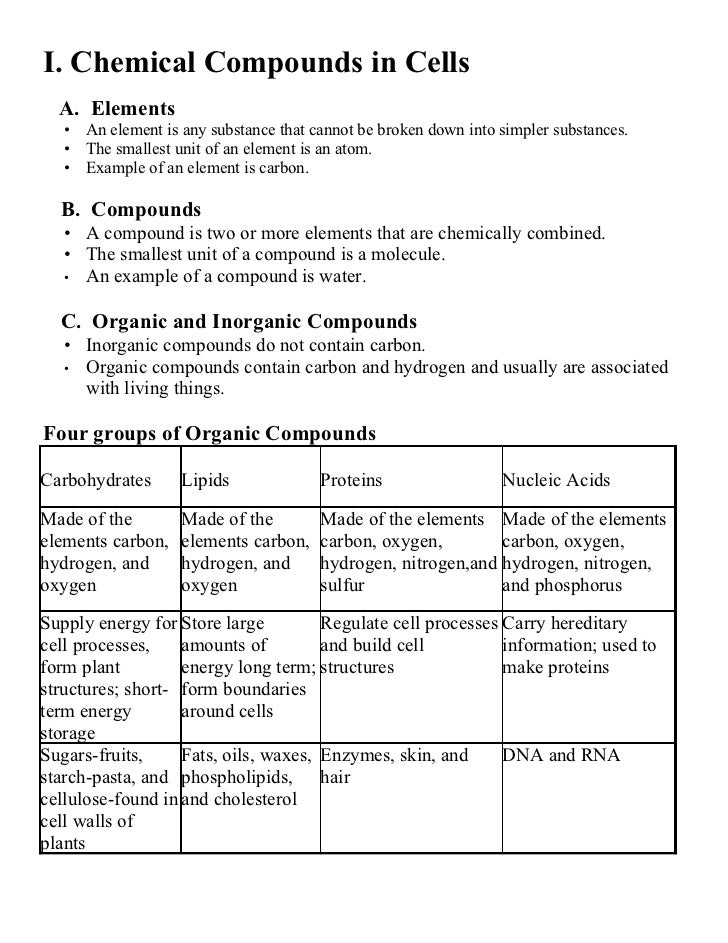


0 Response to "41 2.3 carbon compounds worksheet answers"
Post a Comment